
Based on the 5th from the AQA 9-1 GCSE Chemistry (OUP scheme) C2: The Periodic table chapter. Reactivity is dependent on the classification of an element (metals and non-metals), as they both have differing periodic trends. Standard reduction potentials of aqueous alkali metal cations. During this lesson (C2-5 Explaining trends) GCSE students work through a variety of tasks to develop their understanding of the trends in reactivity in Group 1 and Group 7. Not to miss out, element 102, Nobelium, No, is named in honor of Alfred Nobel, who set aside his vast fortune to establish Nobel Prizes.\). 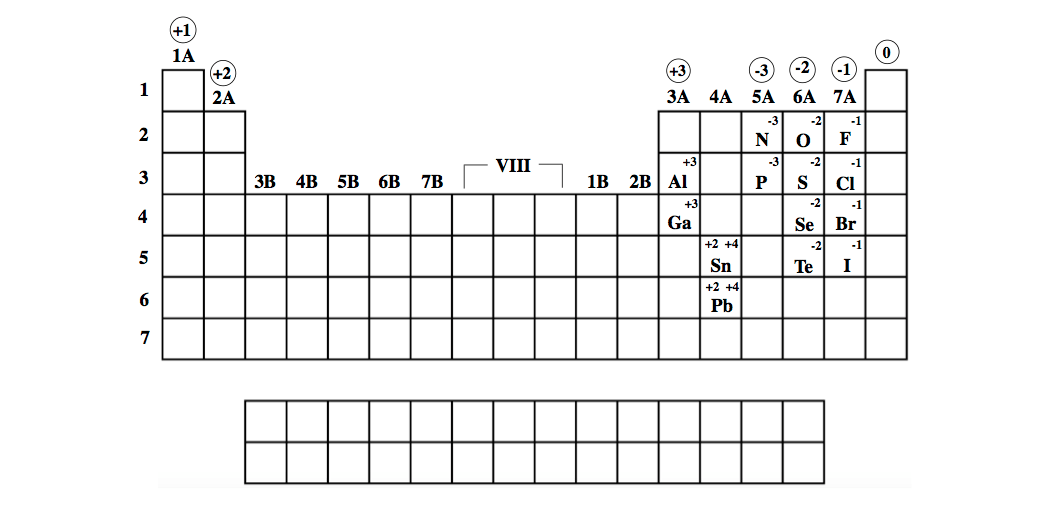
These trends can be predicted merely by examing the periodic table and can be explained and understood by analyzing the electron configurations of the elements. Mendeleev never received a Nobel Prize for his work, but element 101 was named Mendelevium, Md, after him. The periodic table arranges the elements by periodic properties, which are recurring trends in physical and chemical characteristics. For instance they discovered phosphorus when they isolated it from urine. Over time these gaps have gradually been filled in as scientists unearthed new elements. Not only did Mendeleev arrange the elements in the correct way, but he also had the foresight to leave gaps for undiscovered elements. 
Figure 6 6: Periodic Table showing Atomic Radius Trend. This is the opposite trend to that seen in the alkali metals in Group 1 close Group 1 The first vertical column of elements in the periodic table, starting with lithium and ending with francium. As a result, the atomic radius decreases. a diagonal division on the periodic table that divides the metals from the nonmetals and along which lay the metalloids. a family of non-reactive gases (family 18) on the periodic table. The valence electrons are held closer towards the nucleus of the atom. a family within the nonmetals (family 17) on the periodic table. At that time, he had only 50 elements to arrange. This means that the nucleus attracts the electrons more strongly, pulling the atom's shell closer to the nucleus. The alkali metals share similar characteristic chemical. On moving from left to right horizontally in a periodic table, there is an increase in electronegativity. The Pauling scale is used to measure electronegativity. Electronegativity measures the ability of a molecule to gain electrons within a chemical bond. The group 1 metals are lithium, sodium, potassium, rubidium, caesium and francium and they are found in the first column of the periodic table. Some periodic trends and chemical reactivity in the periodic table are as follows. They form alkaline solutions when they react with water. He wrote the properties of the elements on pieces of card and rearranged them until he realised that, by putting them in order of increasing atomic weight, certain properties of elements regularly occurred. The group 1 metals are known as the alkali metals. Then in 1869, a Russian scientist called Dmitri Mendeleev produced one of the first practical periodic tables. Several other attempts were made to group elements together over the coming decades. The earliest attempt to classify the elements was in 1789, when Antoine Lavoisier grouped the elements based on their properties into gases, non-metals, metals and earths. The discovery of other elements followed regularly and soon it became necessary to arrange them in some sort of order. PubChem is a reliable source of chemical information from authoritative sources. 45 Reactivity of Elements of the Periodic Table Novice 1 m Reactivity is a measure of how easily an element will combine with other elements to form compounds. You can look up element names, symbols, atomic masses and more, or test your knowledge with a periodic table game. The lack of chemical reactivity implied by these failures was of significance in the development of theories of atomic structure. In fact, all late 19th- and early 20th-century efforts to prepare chemical compounds of argon failed. However, the first scientific discovery of an element occurred in 1649 when Hennig Brand discovered phosphorous. PubChem Periodic Table of Elements is an interactive tool that lets you explore the properties and trends of chemical elements. These demonstrations show the similarity of the physical and chemical properties of the alkali metals and the trend in reactivity down group 1 of the Periodic Table. This result was significant because fluorine is the most reactive element in the periodic table. 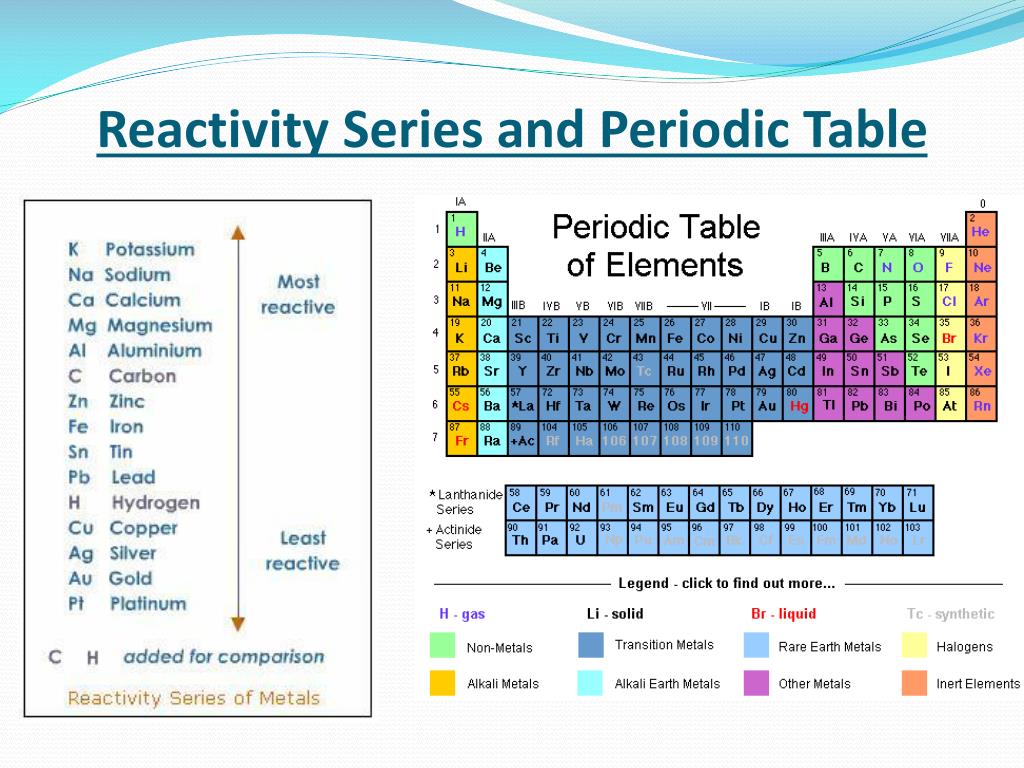
Copper has been used by humans for as much as 7000 years and elements such as gold, silver, tin, lead and mercury have been known for many thousands of years.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
